Background: Approaches to therapy in pediatric and adult cHL have differed historically. Brentuximab vedotin (BV) is efficacious in pediatric patients (pts) with high-risk cHL when combined with chemotherapy and response based involved site radiation therapy (RT) ( Castellino. NEJM 2022). Inhibition of the PD-1 pathway, central to the pathogenesis of cHL, is safe and effective in children with relapsed HL but has not been evaluated in the frontline setting. Led by SWOG, the National Clinical Trials Network (NCTN) conducted the randomized, phase 3 trial S1826 to evaluate nivolumab (N)-AVD vs BV-AVD in pts ages ≥12 years (y) with newly diagnosed Stage 3-4 cHL.
Methods: Eligible pts were randomized 1:1 to either 6 cycles of N-AVD or BV-AVD. At randomization enrollment was stratified by age, International Prognostic Score (IPS), and intent to use RT. Treating site of pts 12-17y, the subject of this subgroup analysis, declared intent to use RT for residual metabolically active lesions on the end of treatment PET per protocol. Recipients of BV-AVD were required to receive G-CSF prophylaxis vs optional with N-AVD. Dexrazoxane was permitted, but not mandated. Response and disease progression was assessed by investigators using the 2014 Lugano Classification. The primary endpoint was PFS; secondary endpoints included overall survival (OS), event-free survival (EFS), and safety.
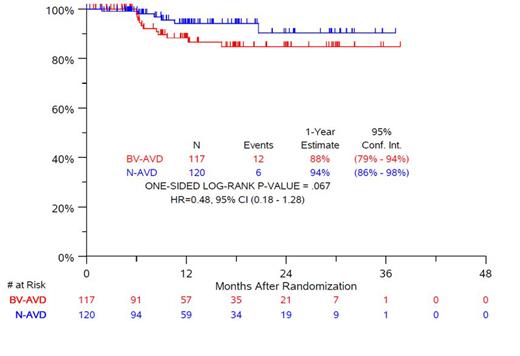
Results: Of 976 eligible pts enrolled (from 7/9/2019 to 10/5/2022): 24.3% (n=237) were 12-17 y and randomized to N-AVD (n=120) or BV-AVD (n=117). Median age was 15.6 y (range, 12-17.9 y), 50% of pts were male, 69% were white, 15% were black, and 18% were Hispanic. 27% had an IPS score 4-7. At the 2nd interim analysis (50% of total PFS events) the SWOG Data and Safety Monitoring Committee recommended reporting the primary results as the primary PFS endpoint crossed the protocol-specified conservative statistical boundary for the full trial. In the pediatric cohort, 6 PFS events were observed after N-AVD compared to 12 events after BV-AVD. At a median follow-up of 12.1 months, the PFS was compared between arms [HR 0.48, 95% CI 0.18-1.28, stratified one-sided logrank p=0.067) in the pediatric cohort; 1 y PFS: N-AVD, 94%, BV-AVD, 88% (Fig.1). There have been no deaths in this age group. Overall use of RT was 0.8% (2/237) and 79% of pediatric pts received dexrazoxane. The rate of grade (gr) ≥ 3 hematologic adverse events (AE) was 45% after N-AVD compared to 41% after BV-AVD, with 3% gr ≥ 3 febrile neutropenia and 1% with sepsis after either regimen, despite differences in GCSF use (59.2% N, 93.2% BV). Overall rates of immune related AEs (irAEs) (any gr) were low: pneumonitis (3.0% N vs 1% BV), and colitis (0% N vs 1% BV). Rash (any gr) was more common in BV-AVD (2% N vs 14% BV).
Hypo/hyperthyroidism (any gr) was higher after N-AVD (2% N vs 0% BV). Rates of transaminitis were similar (ALT elevation: 42% N vs 54% BV). While sensory peripheral neuropathy was more frequent after BV-AVD (sensory: 18% N vs 29% BV; motor: 8% N vs 5% BV), only 1% was > grade 3. Discontinuation of N vs BV therapy occurred in 8.3% vs 2.6% of pediatric pts respectively, compared to 7.9% vs 21.1% of adults.
Conclusions: In early follow-up, N-AVD and BV-AVD are well tolerated and associated with low rates of irAEs in pts ages 12-17 y. With 12.1 mos median follow-up the PFS benefit observed for N-AVD in pediatric pts mirrors that observed in the overall study. RT usage is lower, and cumulative doxorubicin dose is higher than historical pediatric cHL trials. The difference in rate of discontinuation between study arms and by age group needs further evaluation. Longer follow-up is needed to better define the roles of N-AVD and Bv-AVD for AS cHL. S1826 is a model NCTN trial, providing earlier access to novel agents and harmonization of treatment for adolescents and young adults with AS cHL.
Funding: NIH/NCI/NCTN grants U10CA180888 and U10CA180819, U10CA180820, U10CA180821, U10CA180863, U10 CA180886; and Bristol-Myers Squibb. Bv provided by Seagen (Canada Only). Clinical Trial NCT03907488.
OffLabel Disclosure:
Castellino:Bristol Meyers Squibb: Honoraria, Other: Scientific Advisory Committee; SeaGen Inc.: Other: Scientific Advisory Committee - No honoraria, Research Funding. Herrera:BMS: Consultancy, Other: Travel/Accommodations/Expenses, Research Funding; Caribou Biosciences: Consultancy; ADC Therapeutics: Consultancy, Research Funding; Tubulis GmbH: Consultancy; Adicet Bio: Consultancy; Seattle Genetics: Consultancy, Research Funding; AbbVie: Consultancy; Allogene Therapeutics: Consultancy; Merck: Consultancy, Research Funding; Regeneron: Consultancy; Genmab: Consultancy; AstraZeneca/MedImmune: Consultancy; Takeda: Consultancy; Genentech/Roche: Consultancy, Research Funding; Kite, a Gilead Company: Research Funding; Karyopharm Therapeutics: Consultancy; Pfizer: Consultancy; Gilead Sciences: Research Funding; AstraZeneca: Research Funding. Parsons:Seagen: Consultancy. Keller:Merck: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Scientific advisory council. Rutherford:Seagen: Consultancy; Kite: Consultancy; Karyopharm: Consultancy, Research Funding; Genmab: Consultancy; Genentech: Research Funding; Constellation: Research Funding; ADC Therapeutics: Consultancy. Evens:Seattle Genetics: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Morphosys: Consultancy, Honoraria; Epizyme: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Smith:Bristol-Myers Squibb, Gilead, Ono Pharmaceutical: Consultancy, Speakers Bureau. Kelly:Seagen: Other: Scientific Steering Committee; Merck: Other: Scientific Steering Committee.
Nivolumab- not yet approved for frontline disease of Hodgkin Lymphoma in pediatrics

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal